A) "1.0 1010"
B) "10.0"
C) "4.00"
D) "10.0"
E) "4.00"
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a base is dissolved in water, it reacts to give _________ and ________.
A) hydroxide; hydronium
B) hydronium; the conjugate acid
C) hydroxide; the conjugate acid
D) hydronium; the conjugate base
E) hydroxide; the conjugate base
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following reactions best illustrates the reaction of an acid in aqueous solution?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a person hyperventilates, they breathe very quickly, exhaling CO2 more quickly than it can be produced. This reduces the concentration of CO2 in the blood. According to the chemical equation below and LeChatelier's principle, how is the bicarbonate equilibrium affected by the reduction of CO2 in the blood? 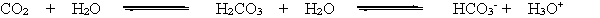
A) The equilibrium is unchanged.
B) More H3O+ is produced.
C) The equilibrium is shifted to the right.
D) The H3O+ concentration decreases.
E) More HCO3 is produced.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following acids is not a strong acid?
A) HCl
B) HI
C) HNO3
D) CH3COOH
E) H2SO4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the acid-base reaction between ammonia and water, which bond is broken in order to give ammonium and hydroxide? 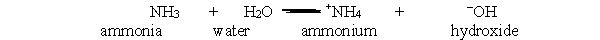
A) H-H
B) N-O
C) N-H
D) O-H
E) All of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The neutralization reaction of potassium hydrogen carbonate (KHCO3) and HI produces what spectator ion?
A) CO2
B) KI
C) H2CO3
D) H2O
E) H+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What do all bases have in common?
A) They contain an -OH group.
B) They contain a nitrogen.
C) They contain an oxygen.
D) They are salts.
E) They contain a nonbonding pair of electrons.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Water can react as both an acid and a base, depending on its environment. Because of this characteristic, water is a(n) _________ molecule.
A) amphoteric
B) autonomous
C) complex
D) reactive
E) conjugated
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a balanced equation for a neutralization reaction?
A) Al(OH) 3(s) + HCl → 3 H2O + AlCl3(aq)
B) Mg(OH) 2(s) + HNO3 → 2 H2O + Mg(NO3) 2(aq)
C) Ca(OH) 2(s) + HNO3 → 2 H2O + Ca(NO3) 2(aq)
D) KOH(s) + HNO3 → H2O + KNO3(aq)
E) 2 Al(OH) 3(s) + HCl → 3 H2O + AlCl3(aq)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a urine sample with a hydronium concentration of 7.9 108?
A) "7.10"
B) "7.10"
C) "6.90"
D) "6.90"
E) "7.00"
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the body, ethylene glycol (antifreeze) is metabolized to a(n)
A) hydrocarbon.
B) diol.
C) ether.
D) toxic organic acid.
E) Ethylene glycol is not metabolized.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The neutralization reaction of potassium hydrogen carbonate (KHCO3) and HI produces what gas?
A) CO2
B) O2
C) H2CO3
D) H2O
E) H+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following figure illustrates the action of a HF and F buffer where the sizes of the boxes are proportional to the concentrations of HF and F in solution. According to this figure, what happens when H3O+ is added to the HF/F buffer? 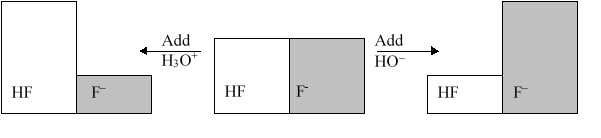
A) The pH decreases.
B) The pH increases.
C) The concentration of HF increases.
D) The concentration of F increases.
E) Nothing happens.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Each circle is a sample of an aqueous acidic or basic solution. Which of the solutions contains a weak acid?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the statements best describes the following reaction? HCOOH + H2O  HCOO + H3O+
HCOO + H3O+
A) This reaction is the dissociation of a strong acid.
B) This reaction is the dissociation of a weak acid.
C) This is the reaction of a strong base.
D) This is the reaction of a weak base.
E) This is not an acid-base reaction.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements best describes what happens when an acid reacts with water? I. The acid donates a proton. II. Water donates a proton. III. Water acts as a base. IV. Hydronium is formed. V. Hydroxide is formed.
A) I only
B) II only
C) I, III, and V
D) I, III, and IV
E) II and V
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lidocaine, a common injectable dental anesthetic, is available as a 1% lidocaine-HCl solution, as shown on the following drug label. Lidocaine-HCl is a(n) 
A) weak acid.
B) base.
C) salt.
D) strong acid.
E) unstable molecule.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following types of molecules and ions is NOT a base?
A) an anion
B) a cation
C) a neutral molecule containing a nonbonding pair of electrons
D) a tertiary nitrogen
E) All of these are bases.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following conditions could cause respiratory acidosis?
A) hyperventilation
B) asthma
C) kidney failure
D) excessive vomiting
E) starvation
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 112
Related Exams