A) 2.90 kJ/mol
B) 3.57 kJ/mol
C) 3.76 kJ/mol
D) 29.7 kJ/mol
E) 31.3 kJ/mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An example of a covalent network solid is
A) diamond.
B) potassium.
C) iodine.
D) sodium chloride.
E) none of these.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
NH3 has a higher boiling point than CH4.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
At a given temperature CCl4 has a weaker surface tension than H2O.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molar enthalpy of vaporization of boron tribromide is 30.5 kJ/mol, and its normal boiling point is 91 C. What is the vapor pressure of BBr3 at 20 C
A) 11.5 torr
B) 311 torr
C) 5.31 torr
D) 143 torr
E) 66.1 torr
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Butter melts over a range of temperatures, rather than with a sharp melting point. Butter is classified as a/an
A) metallic crystal.
B) covalent solid.
C) molecular crystal.
D) amorphous solid.
E) ionic crystal.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The gas phase exists at the point labeled a. 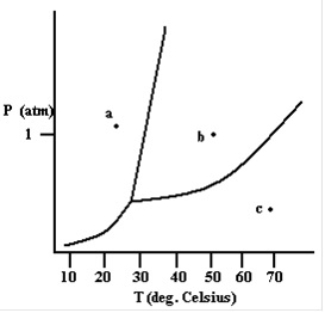
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Ethanol (C2H5 - OH) will have a greater viscosity than ethylene glycol (HO - CH2CH2 - OH) at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of nearest neighbors (atoms that make contact) around each atom in a face-centered cubic lattice of a metal is
A) 2.
B) 4.
C) 6.
D) 8.
E) 12.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Palladium crystallizes in a face-centered cubic unit cell. Its density is 12.0 g/cm3 at 27 C. Calculate the atomic radius of Pd.
A) 154 pm
B) 138 pm
C) 1.95 * 10-8 nm
D) 0.109 nm
E) 1.95 * 10-8 cm
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following liquids and their boiling points, which has the highest vapor pressure at its normal boiling point
A) ethanol, bp = 78 C
B) methanol, bp = 65 C
C) water, bp = 100 C
D) benzene, bp = 80 C
E) The vapor pressure of each of the liquids at its normal boiling point would be the same.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances will have both dispersion forces and dipole-dipole forces
A) HCl
B) BCl3
C) Br2
D) H2
E) CO2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
H2S has stronger intermolecular forces of attraction than H2Se.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The structural form of the element Ge closely resembles the structure of
A) C (diamond) .
B) N (diatomic) .
C) As (tetrahedral) .
D) S (S8 ring) .
E) Kr (monatomic) .
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The intermolecular forces present in C6H6 include which of the following I. dipole-dipole II. ion-dipole III. dispersion IV. hydrogen bonding
A) I, II, III, and IV
B) I and III
C) I, III, and IV
D) I and II
E) III only
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate all the types of intermolecular forces of attraction in CH3OH(l) .
A) Dispersion and Dipole-dipole
B) Dipole-dipole and Ionic
C) Ion-dipole and Hydrogen bonding
D) Hydrogen bonding and Dispersion
E) Dispersion
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the graph of vapor pressure to determine the normal boiling point of O2. 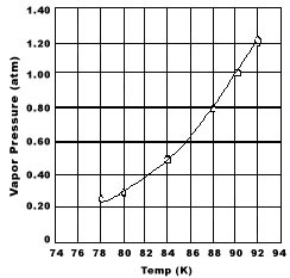
A) 84 K
B) 88 K
C) 90 K
D) 92 K
E) O2 doesn't boil because it is always a gas.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The shape of the water-to-glass meniscus results from the strong adhesive forces between glass and water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following phase changes is endothermic
A) Sublimation
B) Condensation
C) Freezing
D) Deposition
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
At room temperature, honey is more viscous than mustard.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 149
Related Exams