A) 1s22s22p63s23p4 or [Ne]3s23p4
B) 1s22s22p63s2 or [Ne]3s2
C) 1s22s22p63s23p6 or [Ne]3s23p6
D) 1s22s22p63s23p5 or [Ne]3s23p5
E) None of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the ground-state electron configuration of Pb
A) [Xe]6s25d106p2
B) [Xe]6s26d106p2
C) [Xe]6s24f146p2
D) [Xe]6s25f146d106p2
E) [Xe]6s24f145d106p2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If one electron is added to the outer shell of chlorine, it would have the same electron configuration as what element
A) Sulfur
B) Argon
C) Bromine
D) Potassium
E) None of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following electronic transitions is consistent with a decrease in energy
A) From a 5s subshell to a 4d subshell
B) From a 4f subshell to a 5p subshell
C) From a 6s subshell to a 5d subshell
D) From a 5s subshell to a 5p subshell
E) From a 4f subshell to a 6p subshell
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following, if any, is the correct ground state electron configuration for Ni
A) [Ar] 4s23d5 or 1s22s22p63s23p64s23d5
B) [Ar] 4s23d6 or 1s22s22p63s23p64s23d6
C) [Ar] 4s23d7 or 1s22s22p63s23p64s23d7
D) [Ar] 4s23d8 or 1s22s22p63s23p64s23d8
E) None of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An FM radio station broadcasts at a frequency of 101.7 MHz. Calculate the wavelength of the broadcast signal in meters. (c = 2.9979 * 108 m/s)
A) 2.948 m
B) 2.958 m
C) 2.968 m
D) 2.978 m
E) None of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An AM radio station broadcasts at a frequency of 1270 kHz. Calculate the wavelength of the broadcast signal in meters. (c = 2.9979 * 108 m/s)
A) 256 m
B) 236 m
C) 216 m
D) 196 m
E) None of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of radiation that has a frequency of 5.39 * 1014 s-1? (c = 2.9979 * 108 m/s)
A) 1.80 * 10-3 nm
B) 556 nm
C) 618 nm
D) 6180 nm
E) 1.61 * 1023 nm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a ground-state vanadium atom is
A) [Ar]4s24d3.
B) [Ar]4s24p3.
C) [Ar]4s23d3.
D) [Ar]3d5.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following sets of quantum numbers represents an electron with the highest energy
A) n = 2, l = 1, ml = 0, ms = +1/2
B) n = 3, l = 0, ml = 0, ms = -1/2
C) n = 2, l = 0, ml = 0, ms = +1/2
D) n = 3, l = 2, ml = 1, ms = -1/2
E) n = 3, l = 1, ml = 1, ms = +1/2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
There is nothing wrong with the following set of quantum numbers: n = 3, l = 1, ml = 0, ms = +1/2
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electron configuration of an excited state of an oxygen atom
A) 1s22s22p4
B) 1s22s22p5
C) 1s22s22p33s1
D) 1s22s22p6
E) 1s22s22p3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, represents the ground state electron configuration for the selenium atom
A) [Ar] 4s23d104p3 or 1s22s22p63s23p64s23d104p3
B) [Ar] 4s23d103p4 or 1s22s22p63s23p64s23d103p4
C) [Kr] 4s23d104p4 or 1s22s22p63s23p64s23d104p4
D) [Ar] 4s23d104p4 or 1s22s22p63s23p64s23d104p4
E) None of the Above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy in joules of one photon of microwave radiation with a wavelength 0.122 m (c = 2.9979 * 108 m/s; h = 6.626 * 10-34 J.s)
A) 2.70 * 10-43 J
B) 5.43 * 10-33 J
C) 1.63 * 10-24 J
D) 4.07 * 10-10 J
E) 2.46 * 109 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The orbital diagram for a ground-state oxygen atom is 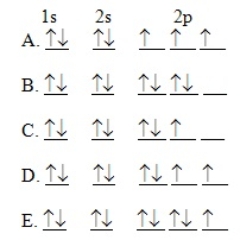
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a hydrogen atom and a helium atom have the same kinetic energy,
A) the wavelength of the hydrogen atom will be about 4 times longer than the wavelength of the helium atom.
B) the wavelength of the hydrogen atom will be about 2 times longer than the wavelength of the helium.
C) the wavelength of the hydrogen atom will be roughly equal to the wavelength of the helium atom.
D) the wavelength of the helium atom will be about 2 times longer than the wavelength of the hydrogen atom.
E) the wavelength of the helium atom will be about 4 times longer than the wavelength of the hydrogen atom.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are allowed in a subshell if l = 2
A) 1
B) 3
C) 5
D) 7
E) 9
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength, in meters, of an alpha particle with a kinetic energy of 8.0 * 10-13 J (Mass of an alpha particle = 4.00150 amu; 1 amu = 1.67 *10-27 kg)
A) 8.4 * 10-15 m
B) 7.4 * 10-15 m
C) 6.4 * 10-15 m
D) 5.4 * 10-15 m
E) None of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the ground-state electron configuration of Ir
A) [Xe]6s25d7
B) [Xe]6s26d7
C) [Xe]6s24f145d7
D) [Xe]6s25f146d7
E) [Xe]4s25f146d7
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second line of the Balmer series occurs at a wavelength of 486.1 nm. What is the energy difference between the initial and final levels of the hydrogen atom in this emission process
A) 2.44 * 1018 J
B) 4.09 * 10-19 J
C) 4.09 * 10-22 J
D) 4.09 * 10-28 J
E) 1.07 * 10-48 J
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 135
Related Exams