Correct Answer

verified
The acidity of a substance can be often ...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Although all bond lengths in benzene are identical,that is not always true for all aromatic compounds.In naphthalene,for instance,the C1-C2 bond is shorter than the C2-C3 bond.Explain,using resonance theory.
Correct Answer

verified
The major resonance structures for napht...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following would you expect to be antiaromatic? 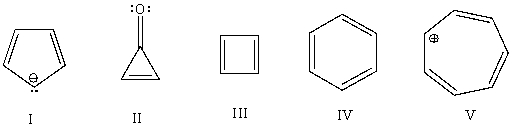
A) I
B) II
C) III
D) IV
E) V
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would you expect to be aromatic? 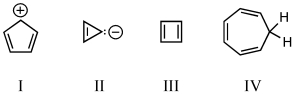

A) I
B) II
C) III
D) IV
E) V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these species is aromatic? 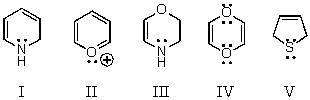
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the only one of these reagents which will react with benzene under the specified conditions?
A) Cl2,FeCl3,heat
B) H2,25°C
C) Br2/CCl4,25°C,dark
D) KMnO4/H2O,25°C
E) H3O+,heat
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about cyclooctatetraene is NOT true?
A) The compound rapidly decolorizes Br2/CCl4 solutions.
B) The compound rapidly decolorizes aqueous solutions of KMnO4.
C) The compound readily adds hydrogen.
D) The compound is nonplanar.
E) The compound is comparable to benzene in stability.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these species is aromatic? 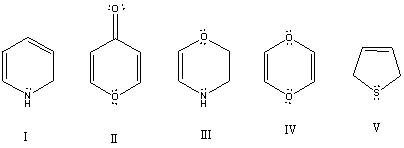
A) I
B) II
C) III
D) IV
E) V
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Briefly explain why the aromatic hydrocarbon azulene,C10H8,possesses a significant dipole moment.Use diagrams as needed to illustrate/clarify your answer.
Correct Answer

verified
Azulene has the bicyclic structure shown...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Draw all significant resonance structures for the cyclopentadienyl anion.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these species is aromatic? 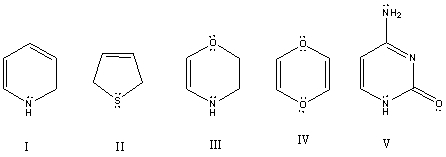
A) I
B) II
C) III
D) IV
E) V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many resonances would be expected in the broad-band decoupled C-13 spectra of: 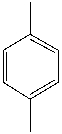
A) 1
B) 2
C) 3
D) 4
E) 5
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these species is aromatic? 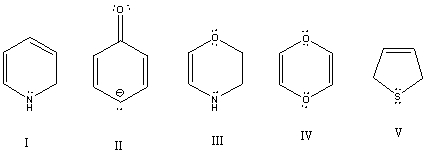
A) I
B) II
C) III
D) IV
E) V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the molecular orbital model of benzene,the six p-orbitals combine to form how many molecular orbitals?
A) 6
B) 5
C) 4
D) 3
E) 2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many different dihydroxybromobenzenes are possible?
A) 8
B) 7
C) 6
D) 5
E) 4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would you expect to be aromatic? 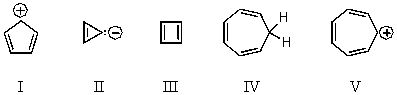
A) I
B) II
C) III
D) IV
E) V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reagent(s) /technique would serve to distinguish between azulene and bicyclo[5.3.0]decane? ![Which reagent(s) /technique would serve to distinguish between azulene and bicyclo[5.3.0]decane? A) Br<sub>2</sub>,CCl<sub>4</sub> B) KMnO<sub>4</sub>,OH <sup>-</sup> ,H<sub>2</sub>O C) NMR Spectroscopy D) NaBH<sub>4</sub>,H<sub>2</sub>O E) Two of these](https://d2lvgg3v3hfg70.cloudfront.net/TB5901/11ea9a02_1b87_d430_8bb6_8fa25154cd0c_TB5901_00.jpg)
A) Br2,CCl4
B) KMnO4,OH - ,H2O
C) NMR Spectroscopy
D) NaBH4,H2O
E) Two of these
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cyclopentadiene is unusually acidic for a hydrocarbon.An explanation for this is the following statement.
A) The carbon atoms of cyclopentadiene are all sp2-hybridized.
B) Cyclopentadiene is aromatic.
C) Removal of a proton from cyclopentadiene yields an aromatic anion.
D) Removal of a hydrogen atom from cyclopentadiene yields a highly stable free radical.
E) Removal of a hydride ion from cyclopentadiene produces an aromatic cation.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these would you expect to have significant resonance stabilization energy? 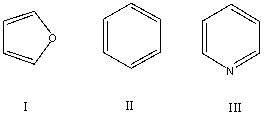
A) I
B) II
C) III
D) All of the above
E) None of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Predict the product of the reaction of one equivalent of hydrochloric acid with the organic
compound shown: 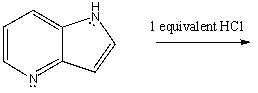
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 151
Related Exams